We have all read posts regarding how to make new batteries from disposed batteries or how to revive old batteries. But, honestly, how many of you knew that you can make a battery with spare change lying around? We bet not many, so here we are to tell you how you can make a battery out of pennies. The battery created from pennies is powerful enough to run an LED light or power your calculator.
 Since they come in various types; the logical question is which pennies in particular are we talking about over here. US Pennies that were manufactured after 1982 will work for this guide. These pennies work because they made up of zinc, in fact, almost 98% composition of these pennies is zinc.
Since they come in various types; the logical question is which pennies in particular are we talking about over here. US Pennies that were manufactured after 1982 will work for this guide. These pennies work because they made up of zinc, in fact, almost 98% composition of these pennies is zinc.
Let’s see how you can make a battery with these pennies by applying some simple engineering knowledge. First off; any individual battery cell is basically a zinc bottom, copper top and these are separated by a material such as paper/cardboard that has been soaked in electrolyte. Now the items you will need are zinc washers that you can get from any local hardware store (they are really cheap).
Now you need to find some cardboard and cut circular pieces of it that are a bit bigger as compared to pennies and soak them in vinegar for about 2 minutes. If you can’t get your hands on vinegar, you can try salt water or lemon juice. They all work for this guide.

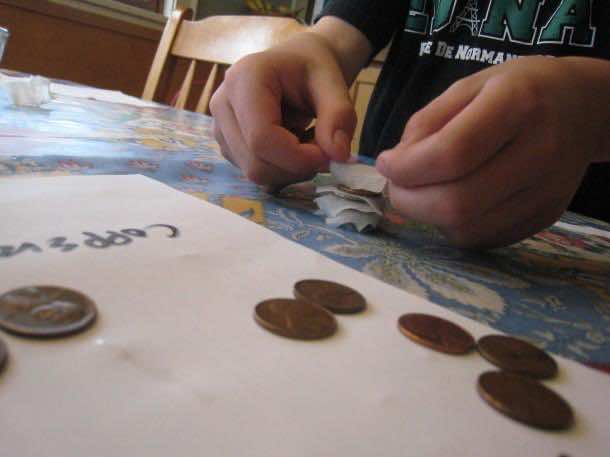 Now, let’s get to it folks; place a aluminum foil on the workspace where you intend to make the battery. Now put a zinc washer on it followed the cardboard piece that was soaked in vinegar and blot dry it with paper towel before placing it on the washer. Next comes the penny that will be placed on top of the cardboard and that’s about it. If you compare this assembly to the cell configuration of a common battery, you would realize that it is the same.
Now, let’s get to it folks; place a aluminum foil on the workspace where you intend to make the battery. Now put a zinc washer on it followed the cardboard piece that was soaked in vinegar and blot dry it with paper towel before placing it on the washer. Next comes the penny that will be placed on top of the cardboard and that’s about it. If you compare this assembly to the cell configuration of a common battery, you would realize that it is the same.

 Each cell will generate about 0.6 Volts and about 700 mA. The zinc bottom will become negative while the copper top becomes positive. The cell will keep on providing power as long as the cardboard has electrolyte. If it stops working, soak the cardboard again and your battery will start working again.
Each cell will generate about 0.6 Volts and about 700 mA. The zinc bottom will become negative while the copper top becomes positive. The cell will keep on providing power as long as the cardboard has electrolyte. If it stops working, soak the cardboard again and your battery will start working again.


 Now if you are looking for an assembly that can generate more power; get ready! Take a hold of 100 grit sandpaper and sand one of the face of the penny exposing the zinc. Follow the same steps but this time around, start stacking pennies on one another with all of them facing the same way. This ensures that zinc top remains positive and copper bottom is negative. By attaching these batteries in series (a total of 10 of them), the voltage can be increased to 6 volts.
Now if you are looking for an assembly that can generate more power; get ready! Take a hold of 100 grit sandpaper and sand one of the face of the penny exposing the zinc. Follow the same steps but this time around, start stacking pennies on one another with all of them facing the same way. This ensures that zinc top remains positive and copper bottom is negative. By attaching these batteries in series (a total of 10 of them), the voltage can be increased to 6 volts.
 For those of you who are wondering how long the LED will last with this 6 volt battery; the one we tried out lasted for about 15 days! Good luck to all those who will be trying this guide out and let us know your experience in comments section. Check out the youtube video below for more:
For those of you who are wondering how long the LED will last with this 6 volt battery; the one we tried out lasted for about 15 days! Good luck to all those who will be trying this guide out and let us know your experience in comments section. Check out the youtube video below for more:



These are really typical types of unintentional removal connected with Vision Unit.
You might try making use of sorting instances to make every the components isolated.
This really is a great way to remain organized. I know my garage area and basement are really a mess.
I am undoubtedly planning on which makes something along these lines.
Thanks, this is a wonderful science project for my kid and my friends kids
can we increase the potential by changing the thickness, surface area of the coins and washers used? can we use any other metal for the same? what if want to increase the current keeping the potential same?
really amazing how money influences the goals. REALLY source full of gain. That is a huge draw in to do you see how this is your life.