Electricity has done wonders to humanity. However, the problem is that most of the energy produced goes wasted in form of heat. Scientists from MIT and Stanford University have found a way to convert battery’s waste heat into more electricity. This indeed is an advancement that could give a whole new dimension to reusable energy sources.

 The concept of converting battery’s waste heat into more rechargeable power is based on the thermogalvanic effect. The principle behind this effect is that temperature has an impact on the voltage of rechargeable units/batteries. The researchers made use of heat, which normally goes wasted to warm an uncharged battery. This warm battery is charged and once it hits the full mark, it is then cooled down. This cooling discharges the battery and raises its voltage. The amazing thing about this battery is that after getting cooled down, it possesses more power than the energy utilized to charge it. This applies to cases where temperature differences are less than 100 degrees Celsius.
The concept of converting battery’s waste heat into more rechargeable power is based on the thermogalvanic effect. The principle behind this effect is that temperature has an impact on the voltage of rechargeable units/batteries. The researchers made use of heat, which normally goes wasted to warm an uncharged battery. This warm battery is charged and once it hits the full mark, it is then cooled down. This cooling discharges the battery and raises its voltage. The amazing thing about this battery is that after getting cooled down, it possesses more power than the energy utilized to charge it. This applies to cases where temperature differences are less than 100 degrees Celsius.
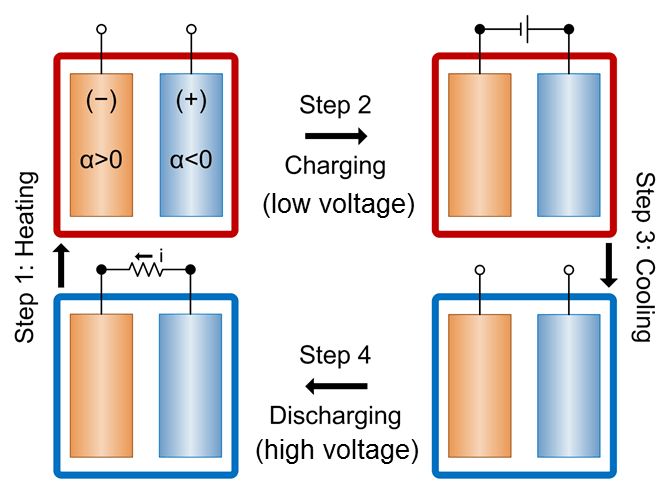
 This new concept has been stated in a paper published in the journal Nature Communications by Yuan Yang and professor Gang Chen at MIT, postdoc Seok Woo Lee and professor Yi Cui at Stanford, and three others. “Virtually all power plants and manufacturing processes, like steel making and refining, release tremendous amounts of low-grade heat to ambient temperatures,” said Cui, an associate professor of materials science and engineering. “Our new battery technology is designed to take advantage of this temperature gradient at the industrial scale.” “To harvest thermal energy, we subject a battery to a four-step process: heating up, charging, cooling down and discharging,” said Lee, a postdoctoral scholar at Stanford and co-lead author of the study. The temperature differences of less than 100 degrees Celsius holds the key in experiments performed. “One-third of all energy consumption in the United States ends up as low-grade heat,” said co-lead author Yang, a postdoc at MIT.
This new concept has been stated in a paper published in the journal Nature Communications by Yuan Yang and professor Gang Chen at MIT, postdoc Seok Woo Lee and professor Yi Cui at Stanford, and three others. “Virtually all power plants and manufacturing processes, like steel making and refining, release tremendous amounts of low-grade heat to ambient temperatures,” said Cui, an associate professor of materials science and engineering. “Our new battery technology is designed to take advantage of this temperature gradient at the industrial scale.” “To harvest thermal energy, we subject a battery to a four-step process: heating up, charging, cooling down and discharging,” said Lee, a postdoctoral scholar at Stanford and co-lead author of the study. The temperature differences of less than 100 degrees Celsius holds the key in experiments performed. “One-third of all energy consumption in the United States ends up as low-grade heat,” said co-lead author Yang, a postdoc at MIT.
 This concept of charging batteries is about 60 years old. Scientists lacked the technological advancements required to prove the concept at that time. Moreover, the required material wasn’t available at that time. “A key advance is using material that was not around at that time for the battery electrodes, as well as advances in the engineering system”, said co-author Chen, a professor of mechanical engineering at MIT. “This technology has the additional advantage of using low-cost, abundant materials and manufacturing processes that are already widely used in the battery industry,” added Lee.
This concept of charging batteries is about 60 years old. Scientists lacked the technological advancements required to prove the concept at that time. Moreover, the required material wasn’t available at that time. “A key advance is using material that was not around at that time for the battery electrodes, as well as advances in the engineering system”, said co-author Chen, a professor of mechanical engineering at MIT. “This technology has the additional advantage of using low-cost, abundant materials and manufacturing processes that are already widely used in the battery industry,” added Lee.
 This system also possesses a few deficiencies as this technology produces lower power densities, which means that the larger batteries become a compulsion. Moreover, the charging process takes quite a bit of time, unlike the conventional battery rechargeable systems. There remains a question mark over the durability of these batteries. Despite everything, this technology possesses a huge amount of potential.
This system also possesses a few deficiencies as this technology produces lower power densities, which means that the larger batteries become a compulsion. Moreover, the charging process takes quite a bit of time, unlike the conventional battery rechargeable systems. There remains a question mark over the durability of these batteries. Despite everything, this technology possesses a huge amount of potential.



i need new technology